D. Medical Gains FDA Clearance for Universal Infusion Set
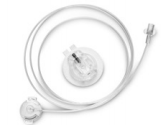
D. Medical's Spring Universal Infusion Set has been cleared by the FDA.
After almost 18 months of review at the FDA, D. Medical’s subsidiary Spring-Set Health Solutions has received clearance for its Spring Universal Infusion Set. More recently, the company also received a medical device license from Health Canada. In addition to its “universal fit,” which applies to any Luer-Lok pump, the Spring Universal Infusion Set has several attractive innovative features, including: a "Detach-Detect" mechanism that alerts users when their sets are detached from the body; a hidden, auto-retractable 28-gauge needle with a trocar tip designed to reduce pain and scarring and to minimize cannula bending potential; and a 360°-connector. By simplfying the insertion process to a one-touch button push, the Spring Universal Infusion Set allows more users to properly insert the sets on their first try. D. Medical intends to make the infusion sets available in the US and Canada in the next few months. --JS







