The Latest European Experiences with Abbott’s Groundbreaking FreeStyle Libre System
By Adam Brown
.JPG)
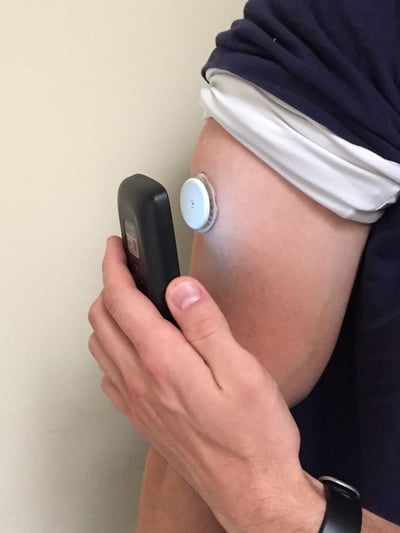
At this year’s ATTD Conference in Paris, a good deal of excitement centered on Abbott’s FreeStyle Libre glucose monitoring system, which launched in Europe last October. The 14-day sensor and touchscreen reader is intended to replace glucose meters, but provide CGM-like information (real-time reading, trend arrow, history). In line with our positive experience wearing the system, the Libre has been a major hit in Europe, and Abbott’s biggest challenge is actually meeting demand. While there weren’t many new tech updates on the Libre at ATTD, we did learn more about the system’s accuracy and how patients and health care providers like it.
FreeStyle Libre is Accurate in Hypoglycemia
New data presented at ATTD suggested that the Libre’s overall rate of accuracy is ~12% vs. a lab analyzer (e.g., if blood glucose is 100 mg/dl on average, the system would be off by about 12 mg/dl on average). This was identical to the real-world accuracy we find vs. a blood glucose meter. It’s remarkably strong considering FreeStyle Libre does not require any blood glucose meter entries (calibrations) to begin reading glucose values – it’s “factory calibrated.” Results also showed that the Libre maintains its accuracy when glucose levels are rapidly changing, as well as during periods of hypoglycemia (glucose levels between 50-80 mg/dl). For comparison, Dexcom’s G4 Platinum with the new Software 505 update has better accuracy at 9.0%, though it requires two calibrations per day; Medtronic’s Enlite sensor comes in at ~14% and requires 2-4 meter calibrations per day.
(3).jpg) First-person testimonies on FreeStyle Libre really put the accuracy into perspective. Dr. Iain Cranston (Portsmouth Hospitals NHS Trust, England) said, “Half my patients have not done a fingerstick in two months. They come to rely on Libre… This should pass as a glucose meter. It meets all the criteria.” We note that the Libre’s guidelines officially recommend that glucose levels be verified in a few cases: when hypoglycemic (low), rapidly changing, or when symptoms don’t match the system’s readings. However, the anecdotal evidence at ATTD suggests that many patients are relying completely on the system. An accuracy trial for Libre is now underway in the US, which could help lead to future US approval.
First-person testimonies on FreeStyle Libre really put the accuracy into perspective. Dr. Iain Cranston (Portsmouth Hospitals NHS Trust, England) said, “Half my patients have not done a fingerstick in two months. They come to rely on Libre… This should pass as a glucose meter. It meets all the criteria.” We note that the Libre’s guidelines officially recommend that glucose levels be verified in a few cases: when hypoglycemic (low), rapidly changing, or when symptoms don’t match the system’s readings. However, the anecdotal evidence at ATTD suggests that many patients are relying completely on the system. An accuracy trial for Libre is now underway in the US, which could help lead to future US approval.
Do Patients Think FreeStyle Libre is Easy to Use?
New user experience data from a 72-person study was presented at ATTD, emphasizing the Libre’s convenience and comfort. Of those studied:
-
100% agreed that the sensor was easy to apply.
-
96% agreed that Libre is comfortable to wear.
-
88% agreed that applying the sensor was less painful than a routine fingerstick (more to the point, we’d add that insertion is only required once every 14 days).
-
86% agreed that the sensor did not get in the way of daily activities.
The Libre is currently only approved for wear on the upper arm, although commentary at ATTD sessions suggests that many patients are experimenting with the sensor’s placement (e.g., buttocks, abdomen), with no noticeable change in accuracy (this is only anecdotal).
 What are the Libre’s Adverse Effects?
What are the Libre’s Adverse Effects?
In the same study, 26 of the 72 volunteers reported some level of discomfort around the sensor insertion site, though all reports were consistent with what would be expected following insertion of a sensor into the skin. There have also been patient reports of mild allergic reactions to the sensor (you can see this on twitter), resulting in skin rashes and irritation, but from our understanding, these instances are rare and most often relatively mild.
Pricing Updates for the Libre
At ATTD, Dr. Cranston approximated that the cost of FreeStyle Libre is equivalent to using ~10 test strips per day. As a reminder, FreeStyle Libre costs 60 euros for each 14-day sensor (120 euros per month), significantly cheaper than CGM. Abbott continues to work on obtaining insurance coverage for Libre (additional studies are still ongoing in type 1 and type 2 diabetes), with expected primary completion dates in May 2015. –AB/AJW







