The Golden Age of the Insulin Pump

The insulin pump has come a long way since the first prototypes were developed in the 1960s. James S. Hirsch explores the riveting history of this integral piece of technology and shares his own experiences with this device.
Part One
We have entered the Golden Age of the insulin pump. It’s been a long time coming.
After decades of disappointment, insulin pumps are living up to the expectations of their early advocates, who believed that these devices could transform the treatment of type 1 diabetes. Thanks to continued improvements in the functionality, safety, and wearability of pumps, they are finally gaining much broader acceptance among consumers.
But this insulin pump story has a twist. While the machines themselves have certainly improved, their Golden Age is due in part to a different technology. Continuous glucose monitors (CGM) can now transmit glucose readings to the pump, which can adjust the insulin delivery automatically to minimize high or low blood sugars. Medtronic, Tandem, and, most recently, Insulet – the three biggest players in the field – now offer these automated insulin delivery systems, which integrate algorithms and wireless technology to support human decision-making.
This combining of the sensor with the pump is also known as a hybrid closed-loop system – “hybrid” because users still have to give the pump essential information, including how much insulin to deliver before meals and how much food will be consumed.
The hope is that the hybrid system is the last step toward a fully closed-loop system, in which the glucose sensor and the pump function like an artificial pancreas, requiring no engagement from the user.
I call this the “Look-Ma-No-Hands” approach to diabetes management, and it remains the Holy Grail of type 1 care.
Even if that goal remains elusive, the pump industry is thriving.
The third quarter of 2021 saw record revenue for insulin pumps – $859 million worldwide, a 22% increase from year-ago sales, and a 68% increase from four years ago, according to Close Concerns, a healthcare information company. Tandem Diabetes Care, whose survival was in doubt five years ago, has now taken the industry by storm; it said that in 2021, it shipped 128,312 pumps, a 41% increase from a year ago. The Insulet Corporation, which makes the Omnipod patch pump for insulin and other injectables, struggled for years, but in 2021, the company surpassed $1 billion in sales for the first time and more than doubled its earnings, to $16.8 million, compared to the year before.
The future appears to be even brighter. Grand View Research, a market research firm, estimates that the global insulin pump market will increase in sales from an estimated $4.6 billion in 2021 to $8.3 billion in 2028.
Reflections from a pioneer
 These buoyant results gratify Freddi Fredrickson, whose personal and professional lives have closely intertwined with the insulin pump.
These buoyant results gratify Freddi Fredrickson, whose personal and professional lives have closely intertwined with the insulin pump.
Fredrickson was a diabetes nurse educator in 1980 when she went on her first pump as part of a study to determine the technology’s impact on long-term diabetes-related complications – the pumps had only been on the market for a couple years, making her a pioneer. She then joined MiniMed in 1983 as the pump company’s seventh employee, and she held numerous jobs over a distinguished 24-year career while working closely with Al Mann, the preeminent visionary in the field.
MiniMed, which was later bought by Medtronic, quickly became the industry leader, where it remains to this day, but it still fell short of its early goals. “We wanted pumps to become the standard of care,” but that didn’t happen, Fredrickson told me in a telephone interview from her home in the Los Angeles area. She said that physicians wouldn’t prescribe a medical device they didn’t understand; educating patients was difficult; and insurers were reluctant to cover an expensive, largely unproven technology.
Fredrickson still uses an insulin pump, making her one the world’s longest continuous users of the device – remarkably, she has worn one for 42 years out of her 61 years with diabetes. Numerous studies, she said, have shown the benefits of pumps, but the glucose sensors have made all the difference as an ideal complement to pump therapy. After all these decades, she can’t quite believe that pump therapy’s original goals may be realized – she called it “amazing.”
“I’m just so surprised that it's finally becoming the standard of care,” she said.
The “Big Blue Brick”
I have my own long history with insulin pumps, mostly positive but also challenging and complicated. I’m now on my fourth different pump from four different pump companies, and I recently began the Tandem Control IQ hybrid system, which I use with my Dexcom G6. While I believe in pumps and continuous glucose monitors, I think their benefits are sometimes misstated. I’ve met pump advocates over the years – I call them pump evangelists – who believe that these machines, now reinforced with continuous glucose monitors, will revolutionize diabetes care.
Call me a cautious evangelist.
 I was diagnosed with type 1 diabetes in 1977 and saw my first pump in 1979 or 1980, when I was a counselor at a summer camp in Missouri for kids with diabetes. A guy showed up to talk about his insulin pump, and the contraption was memorable. It reminded me of a World War II walkie-talkie that I had seen in movies: fancy technology, but large, bulky, and cumbersome; and something that I would never want to hang on my belt. (The prototypes for the insulin pump, developed in the 1960s, were strapped on like a backpack.)
I was diagnosed with type 1 diabetes in 1977 and saw my first pump in 1979 or 1980, when I was a counselor at a summer camp in Missouri for kids with diabetes. A guy showed up to talk about his insulin pump, and the contraption was memorable. It reminded me of a World War II walkie-talkie that I had seen in movies: fancy technology, but large, bulky, and cumbersome; and something that I would never want to hang on my belt. (The prototypes for the insulin pump, developed in the 1960s, were strapped on like a backpack.)
That first pump that I saw was probably the same model that Freddi Fredrickson wore. Its official name was the Autosyringe but, weighing 17 ounces, was nicknamed the “big blue brick.”
The guy who came to our camp had a decent pitch for this oversized medical apparatus. Back then, we used vials of insulin and syringes, which meant two shots a day (that was the standard of care). But with the pump, no more shots! Just strap it on, and you’re good to go.
As I later learned, the pump had other advantages, including the possibility of improved management of blood sugars. At the time, our long-lasting insulin was NPH or Lente, which would take effect after several hours but – depending on which one you used – would peak six, eight, or 12 hours later.
This meant that we were always worried about when our insulin was peaking: Unless we were offsetting that peak with food, our blood sugars would crash. We were, in effect, feeding our insulin instead of our bodies. In retrospect, it was a terrible way to treat diabetes, but it was the best we had.
Replicating a healthy pancreas
Pumps, in theory, solved that problem. The medical term for an insulin pump is “continuous subcutaneous insulin infusion” – a terrible name that only a doctor could love. But it includes a keyword, “continuous.” A healthy pancreas continuously releases a small amount of insulin, day and night – known as background insulin – and then it increases that insulin release when food is consumed.
And that is what a pump does. In its simplest form, it’s a machine that pushes insulin through a tube and into your body, flowing through a cannula that rests beneath your skin. The tubing and cannula make up the insertion set that must be replaced every three days. (The patch pumps deliver insulin without the tube, and they also must be replaced every three days.)
A pump releases a small amount of insulin on a continuous basis, which is the basal rate, and then users can press a few buttons to deliver more insulin before eating food, which is the bolus. Presto! Your pump mimics a healthy pancreas. Daily injections couldn’t do that, at least not when pumps were first introduced, and that led pump advocates to believe these devices would become the standard of care.
Expectations were high. Freddi Fredrickson said that in the early 1980s, there were 23 pump companies worldwide, all attracting premier scientists and engineers, all determined to revolutionize diabetes care.
Confronting obstacles
But whatever the medical advantages of these early pumps, they faced significant hurdles in the marketplace. The most obvious one, as noted, was their size, but even as pumps got smaller, the whole idea of attaching something to your body was foreign – I don’t recall anyone talking about “wearables” in the 1980s and ‘90s. What’s more, technology was something that happened in your home or your workplace, not something you carried with you. (iPhones were introduced in 2007.) Many patients simply didn’t want a device attached to their body and didn’t want to sleep with a small machine in their pajama pocket. (In writing my book “Cheating Destiny,” I interviewed one woman who said her husband wouldn’t have sex with her when she was attached to her pump. She got a divorce and found a more forgiving partner.) Other patients didn’t want a plastic tube snaking out of their pocket or their sleeve, turning an otherwise invisible condition into something quite visible.
Pumps were complicated. They required significant education and training, for clinicians as well as patients. Like any complicated technology, they had glitches and required troubleshooting. They were also expensive. There were safety concerns as well, even with built-in safety features. What if your pump went haywire and started pushing buckets of insulin into your body. A reasonable fear? Probably not. But for all we knew, the thing could kill you.
Another obstacle: doctors had little reason to promote pumps. They were not compensated to train their patients on them, weren’t willing to invest their own time in understanding how these intricate machines worked, and didn’t want their clinics burdened with phone calls when the pumps broke down. To some extent, these hurdles remain.
But perhaps what hurt pumps the most was that their advantages eroded as other therapies improved. The first insulin pens were introduced by Novo Nordisk in 1985, and they were far more convenient and discreet than syringes and vials; and they were followed by next-gen “smart pens,” which made dosing insulin that much easier – all of which hurt the selling proposition of pumps. Then in 2001, a new insulin, glargine, was introduced in the United States by Aventis under the brand name Lantus, and it offered patients nearly 24 hours of a consistent, long-lasting basal insulin. No more peaks! New and better types of basal insulins have since been developed, and they too undermined one of the central advantages of pumps.
While precise numbers are hard to come by, it does not appear that pumps ever exceeded more than 30% of the type 1 market in the United States. They never made significant inroads among those with type 2 who use insulin, and they never became the standard of care. Many pump companies came and went. Even having a great brand name, like the Cozmo or the Asante Snap, couldn’t keep them on the market.
I too had little use for pumps, all the more so because I’m a dedicated technophobe. But I had a surprising change of heart. When you flip your car on a Massachusetts highway because your blood sugar is low, you’ll try something different.
Part Two
There she is
 Insulin pumps received a marketing boost in 1998 when Nicole Johnson wore her MiniMed pump as a contestant in the Miss America pageant. MiniMed sought, and received, FDA approval to create a pink pump, which Johnson proudly showed off. An insulin pump as a sexy fashion item. Who knew? For the swimsuit competition, Johnson wore a two-piece number but did not remove her infusion set, revealing what appeared to be a piece of tape on her stomach, thereby thrusting a flaw, a personal defect, in the faces of the judges. It was an act of bravery and defiance. Johnson was still crowned Miss America, and after that, every little girl with diabetes wanted a pink pump.
Insulin pumps received a marketing boost in 1998 when Nicole Johnson wore her MiniMed pump as a contestant in the Miss America pageant. MiniMed sought, and received, FDA approval to create a pink pump, which Johnson proudly showed off. An insulin pump as a sexy fashion item. Who knew? For the swimsuit competition, Johnson wore a two-piece number but did not remove her infusion set, revealing what appeared to be a piece of tape on her stomach, thereby thrusting a flaw, a personal defect, in the faces of the judges. It was an act of bravery and defiance. Johnson was still crowned Miss America, and after that, every little girl with diabetes wanted a pink pump.
I, on the other hand, continued to resist pumps, but in 2004, after 27 years with type 1 diabetes, I bought my first one. I did it after I had a severe low blood sugar episode while driving on a Massachusetts highway with my 3-year-old son in the backseat. I flipped my car into a ravine. I was lucky – no one was hurt, not even a scratch – but I figured I should try something else, and at the time, that meant trying a pump.
Occlusion errors
I got a black Animus IR 1200, about the size of a pager, and there is no question it was a technological marvel: insulin-on-board information, safety alarms that beeped and vibrated, adjustable basal rates, insulin-to-carb ratios to fine tune dosing, and extended bolusing, which spread the delivery of insulin over time to cover high-fat meals. Compared to insulin injections, the power of the machine was so palpable, the benefits so clear, that I was extra motivated in managing my blood sugars. If perfection was possible, then imperfection was impermissible.
But I had problems with the Animus. I kept getting “occlusion errors” when I bolused, meaning the machine felt resistance from my body when it tried to push through the insulin. When that happened, the pump shut down, and I had to insert a new infusion set. This happened time and again. The company told me there was nothing it could do. The pump just didn’t work for my body, so after about 18 months, I stopped using Animus. (Johnson & Johnson bought Animus in 2005 but discontinued the pump in 2017.)
Next I tried the Roche pump, but the same thing happened: constant occlusion errors, disabling the pump until a new infusion could be inserted. Roche told me that I had the wrong body type – “lean and muscular” – for pumps, which is why they didn’t work for me. I felt like an orphan. I wanted the pumps, but they didn’t want me. So in 2007, I stopped using the Roche as well. (Similar to Animus, Roche stopped selling its insulin pumps in the United States in 2017.)
My fling with insulin pumps appeared to be over, but my doctor told me that Omnipod, whose pods had just reached the market in 2005, had a more forgiving algorithm, meaning they may not shut down when they felt resistance from my body while delivering insulin.
So, I next tried the pods, and they worked. Very few occlusion errors. The technology had then – and still has – both advantages and disadvantages to conventional pumps. Changing the disposable pods was much faster and easier than changing the infusion sets. A patch pump, without tubes, is also less visible (a friend of mine who works as a hospital nurse on a pediatric ward contends with small children who pull her tube).
But delivering insulin through a pod requires a separate device called a Personal Diabetes Manager (PDM), which you carry with you in a black canvas bag, and it doubles as a blood glucose meter. For the past 14 years, whenever I went out to dinner or to a ballgame or most anywhere, I carried my black canvas bag, without which I couldn’t eat food or measure my blood sugar. The president of the United States has the “football” with the nuclear codes. I had my PDM.
Finding a game changer
All these pumps could calculate how much insulin you needed based on your current blood sugar, your expected food intake, and a few other variables. By dialing in your “insulin sensitivity factor,” you could also do correction boluses when your blood sugar was high. But I never used those features. Pumps were evolving to replicate a healthy pancreas, but I was basically my own pancreas. I’ve had type 1 for so long that regardless of the circumstances, I usually know how much insulin I need. Experience beats algorithms.
Even if I didn’t take full advantage of these pumps, all three helped me achieve my principal goal, which was fewer episodes of severe hypoglycemia. Adjusting basal rates over a 24-hour period, as well suspending basal delivery before exercising, gave me far more flexibility and better control.
But the real game changer occurred six or seven years ago when I began using a continuous glucose monitor. (A CGM consists of a glucose sensor, a transmitter, and a reader that tracks glucose values.) Even though my CGM and my pump could not yet communicate with each other, the glucose data gave me information to make more precise decisions, and the CGM’s low-glucose alarms effectively eliminated severe low blood sugars. I used Dexcom and read the values on my smartphone. The CGM wasn’t perfect. Its data was sometimes wrong – I’d use a meter to double check values – and blackout periods occurred in which the transmitter did not communicate with my phone and provided no data.
Still and all, the system worked. The combination of an insulin pump and a CGM gave me A1C values of 6.5% or lower; Time in Range over 70%, and no severe episodes of hypoglycemia.
And that would have been the end of the story, but with diabetes, there is always the unexpected.
Part Three
The stainless-steel solution
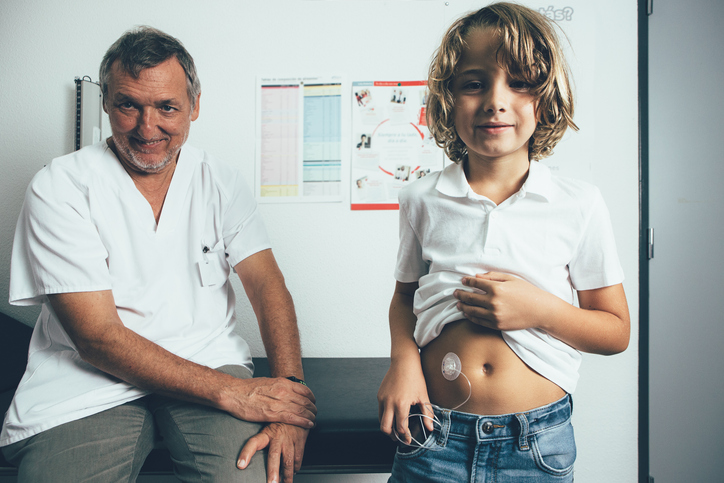 I had been using Omnipod since 2007 and had averted the occlusion errors that had forced me to quit my other pumps, but in 2021, my pods began shutting down with those very same errors. Unlike before, when my pumps shut down while bolusing, my pods just turned off randomly. My doctor concluded that 17 years of wearing pumps had created scarring, or inflammation, beneath the skin. This wasn’t entirely surprising. My Dexcom doesn’t work on my abdomen for the same reason: too much scarring beneath the skin, so I wear my CGM on my arms.
I had been using Omnipod since 2007 and had averted the occlusion errors that had forced me to quit my other pumps, but in 2021, my pods began shutting down with those very same errors. Unlike before, when my pumps shut down while bolusing, my pods just turned off randomly. My doctor concluded that 17 years of wearing pumps had created scarring, or inflammation, beneath the skin. This wasn’t entirely surprising. My Dexcom doesn’t work on my abdomen for the same reason: too much scarring beneath the skin, so I wear my CGM on my arms.
This is a limitation for any wearable. Each body is different. Each body has finite space. And it appeared that my body had finally run its course with pumps.
But my doctor said if I still wanted to use a pump, I could try the Tandem – not because the machine is superior but because I could use it with a stainless-steel infusion set. The pods placed a soft, flexible cannula beneath the skin, and those types of cannulas are commonly used in other pumps. But the steel infusion set might be strong enough to fight through the subcutaneous scarring and deliver the insulin.
So, the t:slim X2, with Control IQ technology, became my fourth insulin pump, and combined with my Dexcom, it was my first sensor-augmented pump. I’ve worn it for about six weeks and will share a few impressions, beginning with the costs.
Without insurance, the machine, which easily fits in the palm of my hand, costs almost $5,000. Meanwhile, the retail price of my Dexcom G6 sensor and transmitter is about $600.
I typically don’t carry much cash. My wristwatch would fetch very little at a pawn shop. My kids tell me that my smartphone is two generations behind. But now with all this wearable diabetes technology, I feel like I’m a high-value target for any robber who can fence medical devices.
I paid $2,000 for my pump, with my insurer covering the rest. Even with insurance, including Medicare and Medicaid, pumps as well as CGMs remain expensive and are beyond the reach of many, and those who do not have good insurance have little chance to afford these devices.
A thinking person’s device
Then there is the complexity of the Tandem. My user guide: 360 pages! That’s worse than my car. Not that there isn’t plenty of help. My trainer for the pump spent two hours with me on a Zoom call, and she also sent me an email with short videos on the many processes and applications of the system. People have posted their own how-to videos on YouTube. The Tandem help desk is a phone call away.
But even for someone like myself, who has worn pumps and CGMs for years and has written about them as well, the amount of information is immense. Any newcomer to these technologies will initially be overwhelmed. These products can be mastered, but let’s acknowledge that these are thinking person’s devices that require a significant investment of time, resources, and intellectual capital to fully exploit.
I don’t blame the companies. It is in their interest to make their products as accessible as possible. But it’s fair to say that the revolution in sensor-augmented pumps will deepen the divide between the haves and have nots in type 1 diabetes. Those of us with financial and educational resources will have new and better ways to care for ourselves, while others will fall behind.
My Tandem trainer did a good job, but she said one thing that made me bristle. She said that in programming your pump, you can give it a name – just type it in, and it flashes on the touch screen. “You can name it whatever you want,” she said. “You know, like ‘Diabetes Sucks.’”
I know she was trying to be funny, and I know that some people have written books titled, “Diabetes Sucks.” But someone without diabetes should never tell someone with it that “diabetes sucks.” And I can’t imagine a more ill-advised suggestion while training someone on a pump. Tandem has invested hundreds of millions of dollars in a device so that diabetes does not suck, and some of us have spent decades, in teaching, writing, researching, or in clinical care, trying to make diabetes a livable condition.
The last thing I want to hear is that our work has been for naught.
If I’m going to bash my trainer for that snafu, I’ll praise her for a brilliant throwaway line. Your Tandem, she said, can be programmed and adjusted for your needs.
“Your body, your pump,” she shrugged.
If I’m running Tandem, I make that my company motto.
A vivid display of life with diabetes
I can see why the Tandem pumps have generated so much buzz and why sales have been so strong. It can do things that no other pump I’ve worn could do. Using algorithms, the Control IQ technology predicts glucose levels 30 minutes ahead and automatically adjusts your insulin – it can decrease your basal rate or give you an additional bolus. Using a t:connect mobile app on my smartphone, I have an hour-by-hour graph of my glucose levels, and the bottom of the graph documents the delivery of insulin: my basal rate, the boluses that I initiate, and the adjustment boluses that the pump does on its own.
The chart is a vivid, humbling display of life with diabetes. Somedays, my glucose graph looks like the rolling plains of Kansas. Other days, it looks like the Six Flags Screaming Eagle roller coaster – I ramp up high, then plunge straight down, then soar too high, then plunge again. It is all there to see, one miscalculation after the next, in food or insulin.
The graph is a reminder that sensor-augmented pumps will not prevent wild spikes in blood sugar. If you eat a plate of spaghetti and don’t bolus enough insulin, the pump on its own cannot “catch up” and keep your blood sugar in range – though it can sound an alarm that you’re about to catapult out of range. The same is true for going too low. If you badly miscalculate your food, insulin, or exercise, you’ll still crash – but you’ll hear alarms on your way down.
Hypoglycemia in surround sound
Nonetheless, the insulin adjustment feature can prevent slow-moving lows. I woke up one morning and saw that while I was sleeping, at around 2:30 a.m., my blood sugar had drifted down and was as low as 74 mg/dL, when my basal insulin shut down. Then my blood sugar gradually drifted upward. I woke up at 95 mg/dL. Amazing.
On another occasion, I was driving home one night, and my blood sugar started dropping to near hypoglycemic levels. I wasn’t feeling anything, but suddenly, my pump started to alarm. Then my smartphone, which is paired to my pump, started buzzing. Hypoglycemia in surround sound! Years ago, I could have killed myself and my son when I went low while driving. Now, I ate a protein bar and was fine.
This is the most important breakthrough for the sensor-augmented pumps. Episodes of severe hypoglycemia have essentially been eliminated – it would take a complete failure of the technology or a massive miscalculation by the user to have such an episode. The importance of this development cannot be overstated. Ever since insulin was discovered in 1921, the principal constraint on its use has been severe hypoglycemia. Physicians have long been reluctant to aggressively prescribe insulin because its misuse could lead to tragic outcomes, even if insulin’s under-use meant patients ran chronically high blood sugars.
Remove severe hypoglycemia as a threat, and you change the course of type 1 diabetes.
Inevitable glitches
Not everything has gone perfectly with my Tandem. Things never do, not with any pump or any technology, though my first mishap was my fault. I live in the Boston area and traveled to New York for several days on business. This was my first time traveling while wearing the Tandem. On my second day, I had to change my infusion set, which comes with three pieces, each in its own package. I was proud that I brought backup packages in case anything went wrong. Alas, I only brought two of the three pieces, rendering the entire pump useless. I also brought one insulin syringe, so for the next 36 hours, I used that syringe to micro-dose insulin every few hours. By the time I got home, the needle was so bent that I thought it was going to snap.
On another trip, I had to fly out of Boston at 5:30 a.m. On my way to the airport at 4:15 a.m., with my wife driving, I gave myself a bolus . . . and the alarm sounded: “occlusion error.” This is Murphy’s Law of diabetes. When something can go wrong, it does, usually at the worst possible time.
I had to change my infusion set at the airport, which is never ideal but at least I remembered all the parts. I stood at a table in a near-empty food court. With all my supplies scattered about, a recorded voice came on the loudspeaker that told travelers to be aware of anything suspicious. “If you see something, say something,” the voice said. I half expected someone to turn me in because it appeared I was strapping an explosive device to my body.
I made it to my plane without incident.
That’s been my only occlusion error with the Tandem, so the infusion set’s steel needle, for now, is working, but it’s not perfect. One time, the needle hit a nerve hours after it had been inserted, and it was it so painful I had to take it out. On another occasion, my blood sugar inexplicably went through the roof. I knew I wasn’t getting my insulin, but the pump sounded no alarms. I feared my $5,000 pump was broken. I called the Tandem help desk, which instructed me to disconnect the tubing from the machine and to bolus insulin. I did, and the machine squirted insulin through the cartridge and into my hand. The machine was fine, but somehow the tubing got twisted and blocked the insulin from entering my body.
A changing culture for insulin pumps
These setbacks are the inevitable tradeoffs for any insulin pump or any diabetes device. Troubleshooting will always be required. Improvisation helps. Persistence and vigilance are required. The only question is whether the advantages outweigh the disadvantages.
In the history of insulin pumps, the answer has been “no” for most people with type 1 – until recently. Now these people are saying yes, and hybrid technology has played a big role. Pumps, combined with continuous sensors, can do more than ever in keeping blood sugars in range and in keeping people safe; and the positive word on these products helps explain their broadening appeal.
Our culture has also changed. Wearable technologies have become common, be it fitness trackers, smart jewelry, body-mounted sensors, or augmented-reality headsets. Most of these are directly related to our physical, mental, or emotional health. So too are insulin pumps. They were once shunned by those of us who were desperate to avoid bringing attention to our medical condition, but now pumps are part of a cool revolution in self-care, just another sexy gadget on our health journey. Not only can we wear pumps with less fear of ostracism but we can showcase them for their wizardry. I speak from experience: insulin pumps fascinate techies and give those of us who can barely program our clock radios an unexpected cachet.
Deepening the conversation
I have my own view on why pumps, combined with continuous sensors, benefit people. I sometimes hear that these devices reduce the burden of care or make managing diabetes easier or more convenient. Maybe for some, but I don’t believe that’s the true value of these technologies.
At its core, diabetes is a continuous conversation with your own body. Every minute of every day, you are trying to figure out what your body is saying and how you should respond. A continuous glucose monitor translates your body’s talking points immediately and clearly, and an insulin pump allows you to respond precisely and preemptively. Can things still go awry? Of course. But these technologies ensure the conversation continues unabated. The attachments on your body do not allow you to think less about diabetes. The exact opposite is true. They force you to think even more about your condition, making the conversation deeper and more meaningful, and that engagement makes all the difference.
As for me, my infusion set’s steel needle is allowing me to wear the Tandem, but I suspect my time is limited. At some point, I fear, the scarring beneath my skin will bottle up these needles and resist the insulin, just as the scarring has done with the other pumps. Nonetheless, I hope to see the fully closed-loop system come to fruition – the artificial pancreas, in all its mechanical glory – but I’m not sure that my storm-weathered body will be a good candidate for it.
No complaints from me. I’ve witnessed the evolution of an industry. Now let’s hope the Golden Age of the insulin pump is the gilded precursor for a better future.
I want to acknowledge Kelly Close, Freddi Fredrickson, Dr. Francine Kaufman, Dr. Irl Hirsch, and Dr. Jay Skyler for helping me with this article.