Abbott Receives European Approval to Launch the Freestyle Libre System – Glucose Monitoring Without Fingersticks
.jpg) Twitter Summary: Abbott receives EU Approval to launch Freestyle Libre – updates from EASD on payment, device accuracy, design, and user experience
Twitter Summary: Abbott receives EU Approval to launch Freestyle Libre – updates from EASD on payment, device accuracy, design, and user experience
On September 3, Abbott announced that it received a CE mark (the European stamp of approval to market and distribute a product) for its Freestyle Libre Flash Glucose Monitoring System. In the upcoming weeks, the product will become available in France, Germany, Italy, the Netherlands, Spain, Sweden and the UK. Abbott intends to bring Freestyle Libre to the US in the future, and a major trial is expected to start before the end of the year. Abbott first introduced this Flash Glucose Monitoring technology a year ago at the EASD 2013 Conference – this system brings an entirely novel technology to the diabetes landscape, eliminating fingersticks to obtain real-time glucose and trend information. The product is intended to be a replacement for traditional blood glucose meters, but is also designed to overcome some of the limitations of CGM (cost, device on the body, need for fingersticks).
.jpg)
The Abbott system includes a tiny glucose sensor (0.2 inches in length, about the thickness of a hair) worn under the skin connected to a water resistant on-body patch the size of a one-dollar coin. The sensor remains inserted for 14 days and does not require fingerstick calibrations (“factory calibrated”); after putting it on the upper arm and waiting one hour, it begins reading glucose and trend information.
To use the system, users take a touchscreen reader device, hold it up to 1.5 inches above the sensor patch, and in less than a second can see their real-time glucose value (e.g., 102 mg/dl), a glucose trend arrow, and a trend graph showing the last eight hours of data. The sensor can be scanned through clothing. The reader device also has a number of nice reports, including a valuable graph that shows time-in-target right on screen. FreeStyle Libre downloads to PC and Mac compatible software, which includes a traffic light approach to identify problematic times of the day. The system is approved for people with both type 1 and type 2 diabetes.
.jpg) FreeStyle Libre is approved for dosing insulin except in three cases: when hypoglycemic, when glucose is changing rapidly, or when symptoms don’t match the system’s readings. In these cases, Abbott recommends confirming the value with a fingerstick. One important difference from current CGM devices (Dexcom G4 Platinum, Medtronic Enlite) is that FreeStyle Libre does not have alarms or alerts, since the sensor data is not sent continuously to the reader device.
FreeStyle Libre is approved for dosing insulin except in three cases: when hypoglycemic, when glucose is changing rapidly, or when symptoms don’t match the system’s readings. In these cases, Abbott recommends confirming the value with a fingerstick. One important difference from current CGM devices (Dexcom G4 Platinum, Medtronic Enlite) is that FreeStyle Libre does not have alarms or alerts, since the sensor data is not sent continuously to the reader device.
At this year’s EASD Conference in September, Abbott held a major symposium on the Freestyle Libre System. Our team attended the event and learned some major updates on the device – our key takeaways are summarized below, including a few updates from their recent press release.
Payment/Reimbursement Updates:.jpg)
-
The touchscreen reader will cost €59.90 (~$77 US). Each 14 day sensor will cost the same.
-
Payment for the system will be out-of-pocket initially, though Abbott is currently enrolling participants for two clinical trials that should help support European reimbursement.
-
People do not need a prescription to purchase the device at online European web sites, which are expected to begin selling the system in the next month.
Freestyle Libre’s Accuracy Updates:
-
Freestyle Libre’s accuracy vs. fingersticks is 11% (e.g., if blood glucose is 100 mg/dl on average, the system would be off by about 11 mg/dl, on average). This accuracy is roughly comparable to the Freestyle Navigator II (12%) and slightly better than the Dexcom G4 Platinum (14%) and the Medtronic Enlite (14%).
Device Design Updates:
-
The patch is only approved for wear on the upper arm.
-
The touchscreen reader has a color screen and a built-in FreeStyle blood glucose meter.
-
The reader’s display has three simple icons: check glucose, history, and settings.
-
User can add notes to each reading and customize the notes.
-
The reader has a micro-USB port to recharge the device, which can connect to a Mac or a PC. Charging the reader for three hours should last up to a week.
-
The sensor patch can store up to eight hours of data. Each scan of the sensor downloads the data to the reader device, which can store up to 90 days of data.
-
Each sensor comes with a simple applicator to apply it to the upper arm. In a demo of the device, this seemed much easier than the insertion process of current CGM products.
User Experience Updates: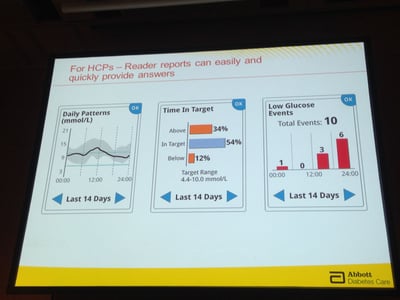
-
According to a user experience study of the Freestyle Libre system, Abbott reported:
-
93% of people agreed that Freestyle Libre is comfortable to wear.
-
91% of people agreed that it is easier to check glucose with this system than with other glucose monitoring systems.
-
83% agreed it was painless to apply the sensor (100% agreed it was either painless or almost painless).
-
96% agreed that Freestyle Libre provides an easy and discreet way to check blood glucose.
-
After seeing a live demonstration of the system, we can attest that it is simple and takes less than five seconds to obtain a glucose reading.
-
From what we have seen, the Freestyle Libre system is easy to use and patient friendly, and its relatively affordable price should make it an attractive option, particularly if reimbursement is approved.
Abbott is currently enrolling participants for two trials of the Freestyle Libre system in Europe. For any EU resident interested in enrolling, please see the clinical trial pages for both the REPLACE trial in type 2 diabetes and the IMPACT trial in type 1. Again, a pivotal study for the Freestyle Libre system is set to begin in the US by the end of 2014. –AJW/AB
For more information on the Freestyle Libre, check out these awesome blog posts:







