What Are Biosimilars?

You may have heard about Semglee and Rezvoglar, popular alternatives for Lantus insulin; these are “biosimilars.” But what is a biosimilar? How is it different from a generic drug? Find answers to these questions and some tips on how to discuss biosimilars with your healthcare provider.
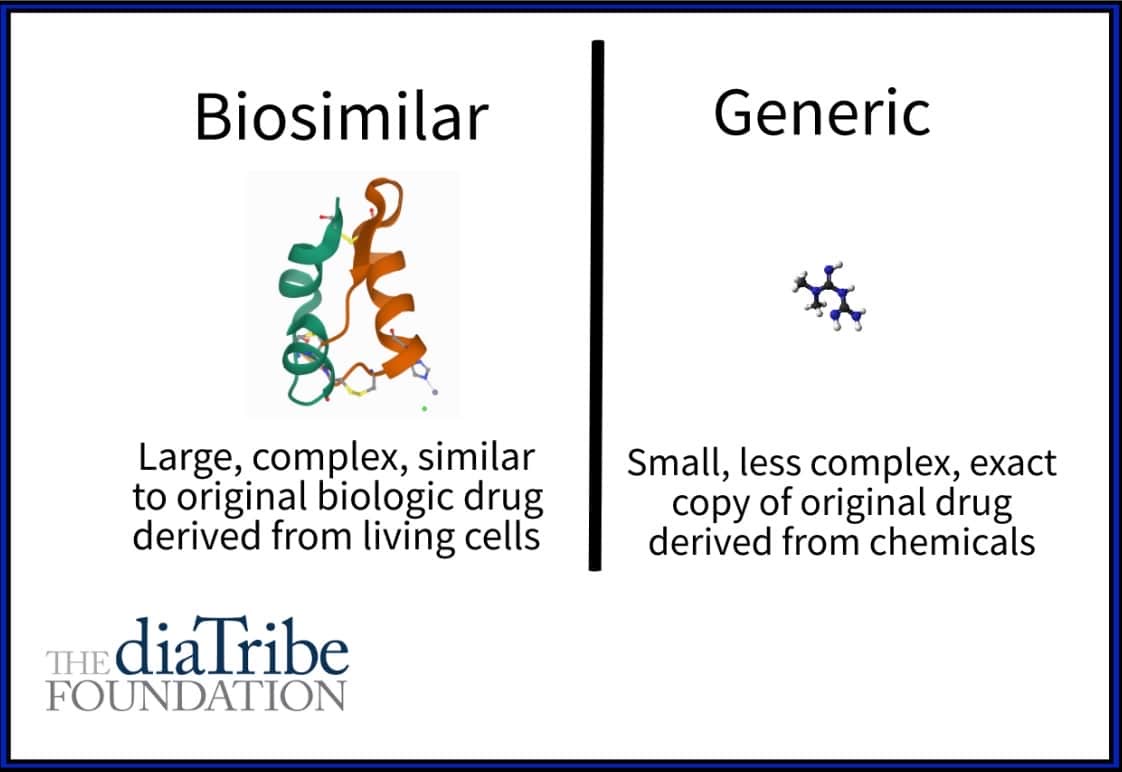
What is the difference between biosimilars and generics?
To understand the difference between biosimilars and generics, it helps to know that there are generally two types of drugs:
-
Biologic drugs – These are large, complex medicines that come from a biological source, such as a living organism. They are challenging and expensive to produce. Examples include hormones like insulin and glucagon.
-
Chemical drugs – These are small, less complex, and as the name suggests, are made from chemicals. These drugs are much easier and cheaper to produce. Examples include metformin and sulfonylureas.
When a pharmaceutical company discovers a new drug, it can obtain a patent on its discovery, meaning that other companies legally cannot manufacture the drug for a certain number of years. During this time, the innovator companies can set high prices for their brand-name drugs, since they are the only ones producing them and need to recoup the costs of drug development.
After the patent expires, any company can produce its own version of the original drug and charge less since they don't have to cover the costs of development and testing the drug. Chemical drugs have simple formulas and can be made exactly like the original product. These are called generic drugs.
Since biologic medicines are derived from living cells, they are far more complex, making it very difficult if not impossible to create an identical replica. Fortunately, advanced biotechnology allows companies to create similar versions of the original biologic medicine that have the same clinical effects. These medications are referred to as biosimilar drugs.
Are biosimilars interchangeable with the original medication?
In terms of safety, a biosimilar medication, once approved, should be considered just as safe as the original product. Dr. Mary Barna-Bridgeman, a clinical professor at the Ernest Mario School of Pharmacy at Rutgers University and internal medicine clinical pharmacist at Robert Wood Johnson University Hospital in New Jersey, said that the FDA requires biosimilars “to undergo rigorous clinical trials to demonstrate safety and efficacy” in order to be approved.
Additionally, biosimilars can also obtain another, more difficult designation from the FDA, which labels it as an “interchangeable biosimilar.” When a biosimilar is classified as interchangeable, it means that a pharmacist can substitute the original brand-name medication for a less expensive alternative without having to consult the prescribing healthcare provider first.
To receive an interchangeable designation, the biosimilar must present additional data from clinical trials to demonstrate that it is as safe and effective in treating a certain disease as the [original] product,” said Barna-Bridgeman. “Without these additional studies, when a new biosimilar receives FDA approval, a prescription must be written specifically for the biosimilar to be used instead of the reference product.”
In July 2021, the FDA approved Semglee to be the first interchangeable biosimilar insulin of Lantus. This meant your pharmacist could provide you with Semglee as a potentially cheaper alternative, even if your healthcare provider wrote you a prescription for Lantus. Based on the state you live in, your pharmacist may or may not have to tell you that they made this substitution when you pick up your prescription.
Rezvoglar, another biosimilar alternative for Lantus, does not have an interchangeable designation, so you would need a prescription specifically for Rezvoglar.
What questions should you ask your pharmacist about biosimilars?
Your pharmacist may provide you with a biosimilar medication for a variety of reasons, the most common reason being potentially a lower cost. “There could be as much as a 30% cost savings with the use of a biosimilar versus certain reference product medicines,” Barna-Bridgeman explained.
If you notice that you received a biosimilar medication as opposed to the familiar brand name, you can ask your pharmacist about its safety, effectiveness, cost, and how the medication works. Take it from Barna-Bridgeman, who said: “Pharmacists are a wealth of information and can relieve any concerns you may have about how it was determined that a biosimilar was right for your use.” Don’t be afraid to ask as many questions as you have at the counter, as your pharmacist is there to help you understand your medications.
The future of biosimilars and diabetes
As a growing number of drug patents are set to expire, there is a large potential for more biosimilar alternatives in the near future. Biosimilar medications, similar to generics, provide additional safe, effective, and potentially more cost-effective options for treating diabetes.