FDA Says No to Zynquista For Now: What’s Next for SGLT Inhibitors in Type 1?

By Emily Fitts and Martin Kurian
Following a close vote from an FDA Advisory Committee in January, the FDA ultimately chose not to approve Zynquista (sotagliflozin), issuing a Complete Response Letter (CRL) instead. The CRL includes the reason for not approving the SGLT-1/2 dual inhibitor for adults with type 1, but the document is not public. The next steps remain unclear, but the CRL could recommend a range of possibilities – from giving the FDA more time to evaluate the application to requiring additional clinical trials.
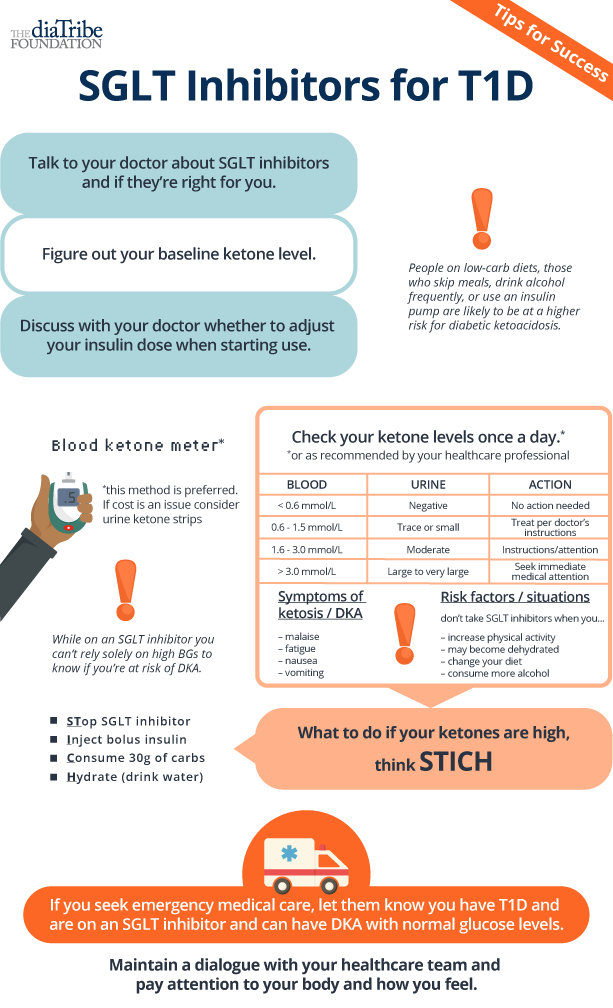
The main concern from the FDA, clinicians, and groups representing people with diabetes is the small but meaningfully increased chance of DKA (diabetic ketoacidosis), which likely contributed to the FDA’s decision not to approve Zynquista without more time and/or information. The CRL affirms FDA’s commitment to ensuring patient safety and may highlight the need for added risk mitigation strategies.
Many experts believe this class of medication fills a large gap in options for people with type 1 diabetes and can be safely taken with the right management and patient support. As one expert, Dr. Satish Garg, Professor of Medicine at the University of Colorado, Denver, said:
“People with type 1 diabetes desperately need another therapy, especially one that goes beyond glucose control. We must now work to help FDA better understand the risk mitigation strategies and beneficial profile of sotagliflozin.”
We received another powerful quote from Dr. Chantal Mathieu, Professor of Medicine, KU Leuven in Belgium:
"I respect the decision of FDA, but do not agree. As a clinician working with people with T1D, I realize every day the unmet needs with our current therapies: not reaching tight enough glycemic control because of increasing risk of hypoglycemia as HbA1c goes down, increasing weight and in particular instability of glucose values, with insufficient time in range. The class of SGLT inhibitors improves all of the above, with impressive increases in time in range of several hours a day! The increased risk of genital infections and the small, but real, increase in DKA is a problem we can overcome with the right education of both patients and medical teams. I am happy that the European Agency has seen this and has proposed a positive decision."
While this is a disappointing outcome near-term, we very much respect FDA’s focus on safety combined with innovation. We are hopeful that it can still be approved at the best time for people with diabetes.
diaTribe has written about SGLT inhibitors for type 1 before – for more background on this topic, see here:
- Using SGLT-2 Inhibitors to Help Manage Type 1 Diabetes
- Making Sense of Ketones with Diabetes
- Two Positive Recommendations by a Regulatory Agency for SGLT Use in Adults with Type 1 Diabetes
- SGLT Inhibitors for Type 1: What to Know
- SGLT Inhibitors for Type 1: Tips for Success Infographic