The Biggest News in Diabetes Technology from ATTD 2020 – A Deep Dive on CGM and more!
By Jimmy McDermottEliza Skoler
 By Jimmy McDermott, Eliza Skoler, Divya Gopisetty, Emily Fitts, Frida Velcani, Kelly Close, and Albert Cai
By Jimmy McDermott, Eliza Skoler, Divya Gopisetty, Emily Fitts, Frida Velcani, Kelly Close, and Albert Cai
The diaTribe team is on the ground in Madrid, Spain to cover the latest news in diabetes technology from ATTD. Here are our top highlights from the conference so far ...!
The 13th annual Advanced Technologies and Treatments in Diabetes (ATTD) conference brought together many great minds in diabetes in Madrid, Spain. With nearly 3,800 attendees from more than 81 countries, ATTD continues to grow dramatically; while it has been since the start in Prague in 2008 one of the most impactful meetings in diabetes, it is now considered one of the top three in the world. Why? It shows the powerful momentum behind diabetes technology in so many respects, from CGM and BGM to automated insulin delivery (AID) to smart pens to new therapies to new learnings in behavioral medicine. Here are some of diaTribe’s key highlights from the conference – we’ve focused on learnings about CGM and the closed loop and will be back with even more after the conference concludes!
Click to jump to a section!
1. Dr. Rich Bergenstal shares tips for understanding CGM data
2. Data shows use of CGM features is linked to increased time in range
4. Results show benefits of DIY closed-loop systems: lower A1C and more time in range using Loop
7. MiniMed 670G data shows time in range increases with Auto Mode
8. Tandem’s Control-IQ increases time in range in children
1. Dr. Rich Bergenstal shares tips for understanding CGM data
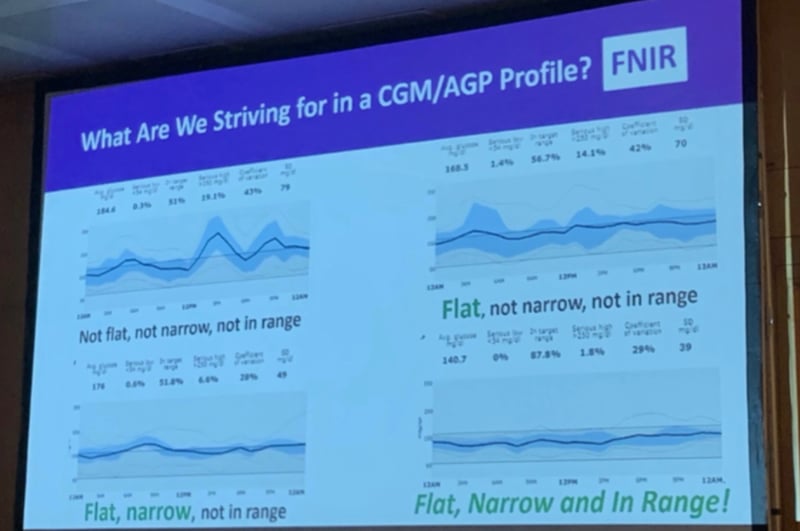
Dr. Rich Bergenstal from the International Diabetes Center gave tips for interpreting continuous glucose monitor (CGM) data. The audience responded enthusiastically when Dr. Bergenstal explained “FNIR” (Flat, Narrow, In-Range) to describe “ideal” blood sugar: high time in range and flat glucose levels with few ups and downs. To learn more about FNIR, check out our resource page on time in range. Dr. Bergenstal shared a few more catchphrases:
-
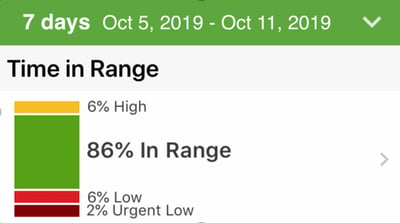 MGLR: More Green, Less Red; refers to the stacked time in range bars (pictured right).
MGLR: More Green, Less Red; refers to the stacked time in range bars (pictured right). -
“Thinking fast and slow” – this refers to making “fast”, in-the-moment medication or behavioral changes based on real-time CGM numbers, and “slow” analyses of blood sugar data to understand and act on trends over time. It also recalls the famous prize-winning book from 2011.
-
STAR: Steady, Tight, And in-Range; this is another one we’ve heard lately, and even though it wasn’t in Dr. Bergenstal’s presentation, it’s also very aspirational, just like FNIR!
2. Data shows use of CGM features is linked to increased time in range
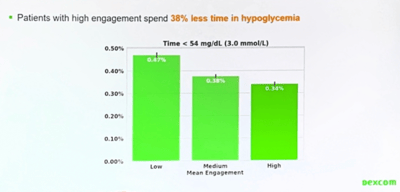
Dexcom presented real-world data from G6 CGM users showing that people who were most engaged with G6 features (such as alerts, the share-follow platform, and Dexcom Clarity) showed significant improvements in their blood glucose management. These highly-engaged users had the highest time in range and spent 38% less time each day in hypoglycemia (pictured right).
Here are the G6 features that Dexcom highlighted:
-
Alert Settings: Users can set their own target blood glucose range and the G6 will automatically notify them if their glucose levels cross the high or low threshold. This means that users don’t have to constantly self-monitor their blood sugar; 55% of G6 users used this feature to customized their settings.
-
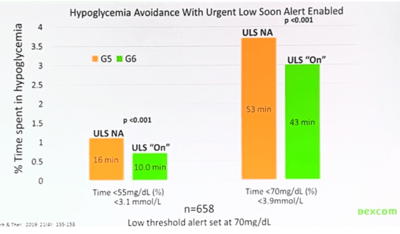 Urgent Low Soon (ULS) alert: This predicts and notifies a user 20 minutes before they cross their low blood glucose threshold. Real-world user data shows that people using ULS spent less time in hypoglycemia (six minutes less below 55 mg/dl and ten minutes less below 70 mg/dl).
Urgent Low Soon (ULS) alert: This predicts and notifies a user 20 minutes before they cross their low blood glucose threshold. Real-world user data shows that people using ULS spent less time in hypoglycemia (six minutes less below 55 mg/dl and ten minutes less below 70 mg/dl). -
Dexcom Follow: People with diabetes can share CGM data with their family, friends, and care partners to help monitor glucose levels (with the iPhone app or Android app). This feature was especially helpful among children, whose time in range increased proportionally to the number of people able to view their data.
-
Dexcom Clarity: Weekly reports allow people with diabetes and healthcare professionals to see blood glucose trends and develop insights into their diabetes management (with the iPhone app or Android app). Users can receive encouraging notifications when they achieve their blood glucose goals. The data shows that more understanding of how one’s glucose levels change over time correlates with better outcomes: using weekly Clarity reports led to an increase in time in range by 9%. Additionally, in a study of 26,000 people, those who used Clarity with mobile notifications had 64% time in range, compared to 52% time in range for people who only used the CGM. Learn more about Clarity here!
3. Dexcom announces CE-Mark for G6 in pregnancy; available in UK “starting spring 2020”
Dexcom announced European approval (formally called the “CE Mark”) for its G6 CGM for use during pregnancy for women living with type 1, type 2, or gestational diabetes. This is so exciting! The official “label” is set to launch in spring of 2020. The G6 now joins Abbott’s FreeStyle Libre as approved CGM for pregnant women in Europe – as a reminder, the FreeStyle Libre 2 has alarms (better for pregnancy), and has been approved in Europe for some time. While no CGM is yet approved for pregnant women in the US (though many are using it off-label), the European announcement should help to increase awareness and education around the world. Every pregnant woman in the world with diabetes or pre-diabetes should have CGM in the opinion of many experts (virtually all the ones we know).
-
Mt. Sinai’s Dr. Carol Levy shared compelling data from the T1D Exchange around CGM in pregnancy. Comparing 2010-2013 to 2016-2018, self-reported CGM use in pregnant women increased from 35% to 65% and average A1C in pregnant women dropped from 6.9% to 6.6%. We bet if time in range data were available, we’d see a big increase.
-
Dr. Levy showed the outcomes of 50 women who used Dexcom CGM during pregnancy. 93% of these women recorded no diabetic ketoacidosis (DKA) or severe hypoglycemia. Typically, about 50% of women with gestational diabetes have a baby with a high birth weight. This percentage was 12% in the group of women using CGM in this trial.
-
As we’ve seen at previous conferences, data supporting CGM during pregnancy is overwhelmingly positive. The CONCEPTT trial examined Medtronic’s Guardian CGM in pregnant women and showed many encouraging results, including:
-
Reduced birth weight of the baby
-
100 more minutes per day in target glucose range for the pregnant mother
-
72 fewer minutes per day in hyperglycemia for the pregnant mother
-
Experts believe that even these encouraging results underestimate the impact of CGM on positive health outcomes for pregnant women and their children. Broadly speaking, pregnancy can cause multiple challenges to diabetes management that CGM can help ease. Pregnant women experience more blood glucose variability with increased insulin resistance and are at a greater risk of hypoglycemia. Learn more here about gestational diabetes.
4. Results show benefits of DIY closed-loop systems: lower A1C and more time in range using Loop
An observational study on Loop, a do-it-yourself (DIY) closed-loop system, found that Loop improved diabetes management significantly. The study followed people using an automated insulin delivery (AID) system, continuous glucose monitor (CGM) readings, and a communications bridge device, called “RileyLink.”
New users (people in the study who had never used this closed-loop system before) showed:
-
An A1C reduction from 6.8% to 6.5% after three months and to 6.4% after six months – and if you think that is not very much, think again!
-
An increase in time in range from 68% to 73%, which is more than one hour per day spent in-range! These time in range benefits occurred in the first month of closed-loop use and were constant throughout the rest of the study.
-
Benefits in A1C and time in range across all age groups.
-
Improvements in user-reported outcomes, including measures of diabetes management distress, sleep quality, and fear of hypoglycemia. Not too many details were shared on this part and we look so forward to learning more!
It is important to note that people in the study were classified as having “well-managed” diabetes at the start of the study, meaning they had relatively low A1Cs (6.8% baseline) and were close to meeting time in range goals, and came from high education levels and socioeconomic status backgrounds. This underscores the need to improve access to closed-loop systems for broader groups of people with diabetes.
Looking ahead, the study will end on March 31, 2020 and all participants will complete a six-month follow-up, so full results will not be available until later this year.
5. WISDM Trial: CGM helps older adults with type 1 diabetes reduce hypoglycemia and improve time in range
The WISDM study examined the impact of continuous glucose monitor (CGM) use in people with type 1 diabetes above the age of 60. Participants used either the Dexcom G5 CGM or standard blood glucose meters (BGM). After six months, the CGM group spent less time in hypoglycemia (with blood glucose below 70 mg/dl) and two more hours per day in-range (70-180 mg/dl), compared to the BGM group. There was also a greater A1C reduction in the CGM group (0.3% decrease compared to no change in the BGM group). Importantly, people using CGM reported significantly fewer severe hypoglycemia events (defined as requiring assistance of another person). These benefits were seen whether people were using insulin pumps or multiple daily injections (MDI).
For the next six months of the study, the individuals using standard blood glucose meters switched over to CGM. New data shows that the people who switched to CGM spent significantly less time in hypoglycemia and more time in range. The CGM-only group maintained the outcomes from the six-month mark, which was also positive to see.
The WISDM study continues to show that CGM is as important, desired, and effective for older adults as it is for younger adults. Indeed, participants who used CGM in this study reported using it 95% of the time. As CGM becomes easier to use and more affordable in the coming years, we look for this valuable technology to be adopted by more people of all ages and backgrounds. That can’t come soon enough!
6. Increased FreeStyle Libre scanning frequency is associated with reduced time with both high and low blood sugar
Dr. Ramzi Ajjan of University of Leeds presented data across many countries and regions showing that more FreeStyle Libre scans each day was associated with reduced time in both hyperglycemia (high blood sugar) and hypoglycemia (low blood sugar).
Unlike traditional CGM, FreeStyle Libre does not continuously send real-time glucose data to the reader; instead, the sensor patch must be “scanned” with the reader to get the real-time glucose value, trend arrow, and trend graph. By scanning more frequently, people are viewing their blood sugar data in real-time more often. This allows them to make adjustments to avoid highs and lows, whether by introducing a new habit or working to change a longstanding behavior.
The data shows that as the number of scans per day increased, time spent in hyperglycemia and hypoglycemia decreased. For example, in the UK, the lowest number of scans (about five per day) resulted in 44% time in hyperglycemia, which fell to 24% time in hyperglycemia with the higher number of scans (about 45 per day – obviously on the high end, but you get the idea!). Find more data here.
7. MiniMed 670G data shows time in range increases with Auto Mode
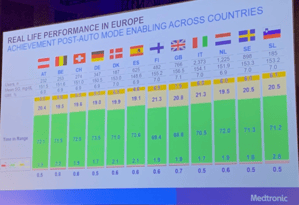 Medtronic presented data from 7,847 people with diabetes across Europe who used the Auto Mode function of Medtronic’s MiniMed 670G AID system. Among 3,139 670G users, average time in range increased from 62% to 71% before and after using Auto Mode – that’s 2.3 hours more time in-range each day!
Medtronic presented data from 7,847 people with diabetes across Europe who used the Auto Mode function of Medtronic’s MiniMed 670G AID system. Among 3,139 670G users, average time in range increased from 62% to 71% before and after using Auto Mode – that’s 2.3 hours more time in-range each day!
The increase in time in range corresponded with a drop in time spent in hypoglycemia and hyperglycemia. These users spent just 1.8% of their time with blood glucose levels under 70 mg/dl, and 0.6% of their time with blood glucose levels under 54 mg/dl. On the hyperglycemia side, users spent 21% of their time with blood glucose levels over 180 mg/dl, and 6% of their time with blood glucose levels over 250 mg/dl. Time in range improvements were seen one month after enabling Auto Mode.
8. Control-IQ increases time in range in children ages 6-13
Dr. R. Paul Wadwa of the Barbara Davis Center for Diabetes presented highly anticipated results from the DCLP-5 trial of Control-IQ in children ages 6-13 years old. Over 16 weeks, time in range increased from:
-
53% to 67% in the group using Control-IQ
-
51% to 55% in the group using a pump and CGM, without Control-IQ.
This means that the children using Control-IQ spent about two and a half more hours each day in their target blood sugar range, compared to the kids using a standard pump and CGM.
Like the adult trial of Control-IQ, most of the increase in time in range occurred at night. At night, the Control-IQ group reached an astounding 80% time in range compared to 54% in the standard pump and CGM group. Overall, time spent above 180 mg/dl was 31% for the Control-IQ group, compared to 43% in the standard pump and CGM group. Time below 70 mg/dl was not changed in either group. There were no cases of diabetic ketoacidosis (DKA) or severe hypoglycemia in the trial.
Dr. Boris Kovatchev from the University of Virginia gave a more detailed comparison of the adult and child Control-IQ trials in the slide below:
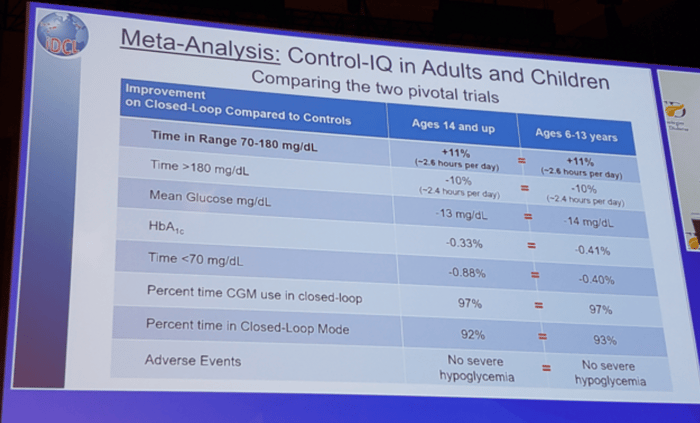
Control-IQ was cleared in December for people ages 14 years and older. Control-IQ is expected to be submitted for approval in children in early 2020.
Thank you so much to the conference organizers Professors Moshe Philip and Tadej Battelino – what an outstanding gathering, and much appreciation for making so many people with diabetes and advocates feel so welcome. You can see so much more on the ATTD Facebook homepage, including the compelling opening ceremony with keynote speaker Dr. Jay Skyler giving a moving talk about technology past and present, as well as a video of talented Spanish musicians and dancers to close out the first night! The live stream was also much appreciated. Thank you so much to technology watcher Albert Cai and his team for their brilliant help in identifying key themes at this memorable conference. We’ll be back with more learnings on therapies, behavior, insulin and more …
This article is part of a series on time in range.
The diaTribe Foundation, in concert with the Time in Range Coalition, is committed to helping people with diabetes and their caregivers understand time in range to maximize patients' health. Learn more about the Time in Range Coalition here.








