Introducing Beta Bionics: Bringing the iLet Bionic Pancreas to Market

By Adam Brown, Alexander Wolf, and Kelly Close
Breaking news today from Boston, where Dr. Ed Damiano of the Bionic Pancreas team disclosed the launch of “Beta Bionics” – a new “public benefit corporation” to commercialize the team’s fully integrated iLet Bionic Pancreas device to automate delivery of insulin and glucagon. Lilly has also invested $5 million in Beta Bionics, a major vote of confidence in the team. The company’s website went live today at betabionics.com.
Beta Bionics will take the Bionic Pancreas platform developed in the academic research setting, integrate it into a single medical device (the iLet), and obtain FDA approval to bring it to people with type 1 diabetes. Dr. Damiano will serve as the company’s President/CEO, and we were thrilled to see that Children with Diabetes founder Jeff Hitchcock is on the Board of Directors – a great way to bring in additional patient perspective! Lilly’s Global Brand Development Leader, Deirdre Ibsen, will also join Beta Bionics’ Board. The unique public benefit corporation approach has some big advantages for people with diabetes – see below.
The timing remains the same as we mentioned in February: a small study later this year to make sure the iLet works like the previous research platform (a “bridging study”), followed by a large pivotal study in mid-2017 testing both insulin+glucagon and insulin-only versions of the automated system. The hope is still to launch the Bionic Pancreas with insulin-only first, with a potential FDA approval in late 2018. The iLet device is designed to function with just the insulin chamber operating, and glucagon could easily be added later once a stable version is approved.
The iLet has also improved since last July’s prototype unveiling at Friends for Life 2015. The system consists of a dual-chamber pump (insulin + glucagon), a Tidepool-designed user interface, and a built-in control algorithm integration that receives CGM information from a separately worn Dexcom sensor. The team has now built a third generation iLet that will be used in upcoming clinical trials; iLet 3 is ~33% smaller than the prototype (it can now fit in a pocket) and has a glass touchscreen for a much better user experience (see picture above). The system will use a single custom infusion set with separate lines for insulin and glucagon.
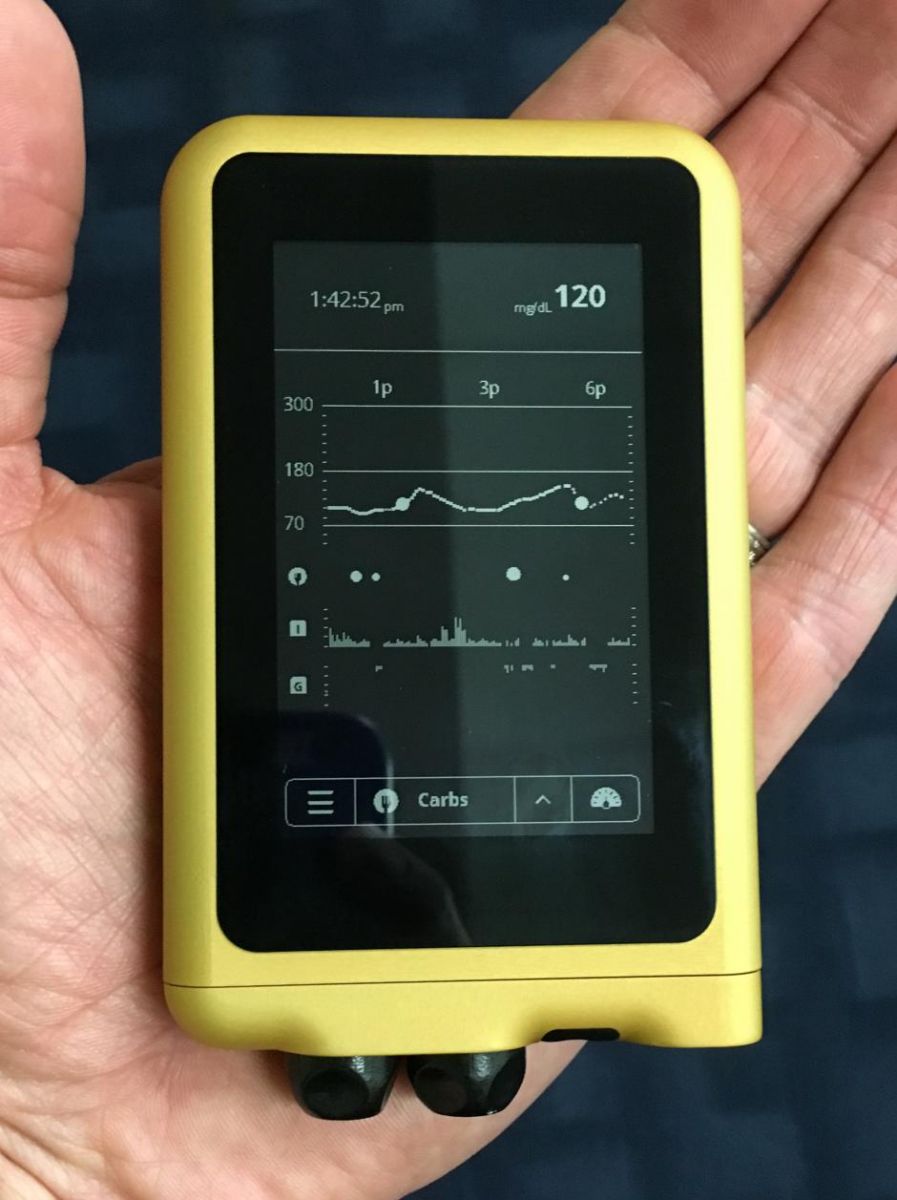 A recently completed eight-hour study with 10 participants tested the iLet in humans for the first time. Results were positive, but did uncover an issue with the device’s custom dual-hormone infusion set. Dr. Damiano assured us that the team has a fix for this problem, and following an FDA discussion, will repeat this exploratory study in the coming months before the bridging study later this year.
A recently completed eight-hour study with 10 participants tested the iLet in humans for the first time. Results were positive, but did uncover an issue with the device’s custom dual-hormone infusion set. Dr. Damiano assured us that the team has a fix for this problem, and following an FDA discussion, will repeat this exploratory study in the coming months before the bridging study later this year.
We spoke with Dr. Damiano this week to learn more about how Beta Bionics will transition the iLet from academic research to a commercial product for people with type 1 diabetes.
Why a Public Benefit Corporation and what does that mean?
The Bionic Pancreas was developed in an academic setting at Boston University and Mass General Hospital, though a company ultimately needs to secure FDA approval and sell it. Instead of giving the iLet to a pump company to bring to market, Dr. Damiano has ambitiously formed Beta Bionics as a dedicated public benefit corporation. Beta Bionics has exclusively licensed all of the Bionic Pancreas-related IP from BU.
A public benefit corporation is a relatively new type of company that leverages the advantages of both for-profit and non-profit organizations. Beta Bionics will be able to raise money like a for-profit company (e.g., from investors), though it will be extremely mission-driven like a non-profit (e.g., JDRF). Traditional corporations have a duty to return money to investors, but as a public benefit corporation, Beta Bionics can prioritize its mission above all else: bring the iLet to as many people with type 1 diabetes as possible. This will ensure the company makes decisions in the best interests of the type 1 diabetes community rather than simply maximizing profits. The public benefit corporation structure should also make fundraising easier, since the team won’t have to rely on just donations. We love this idea!
 As an aside – the logo combines the Greek letters β (beta) and α (alpha) to reflect the dual-hormone design (insulin and glucagon from the beta and alpha cells). Notably, the blue “o” has the three stations of the closed loop paradigm (CGM, pump, algorithm) superimposed on the blue international symbol for diabetes.
As an aside – the logo combines the Greek letters β (beta) and α (alpha) to reflect the dual-hormone design (insulin and glucagon from the beta and alpha cells). Notably, the blue “o” has the three stations of the closed loop paradigm (CGM, pump, algorithm) superimposed on the blue international symbol for diabetes.
Lilly’s investment – a major vote of confidence in automated insulin delivery
Lilly’s $5 million investment is a major move for the pharmaceutical company, who is increasingly moving into diabetes technology in addition to its extensive background making insulin and other diabetes drugs. Lilly’s Deirdre Ibsen shared lots of excitement in our interview: “This investment aligns so well with the strategic direction we [Lilly] are moving in: the convergence of technology and biotechnology.” Indeed, this investment adds on to Lilly’s existing technology partnerships: working with design firm IDEO to design a better injection pen for Trulicity, partnering with Insulet to make a special concentrated insulin OmniPod pump (U200 and U500), and investing in Companion Medical to make a Bluetooth-enabled insulin pen. Lilly also has a new Cambridge innovation center focused on device innovation.
“The Bionic Pancreas is a platform that makes a great drug better,” shared Dr. Damiano in our interview. Past studies of automated insulin delivery have repeatedly shown that insulin needs vary dramatically from day-to day and night-to-night – people with type 1 might require 10 units of insulin on Monday night, seven units on Tuesday, and 13 units on Wednesday. Systems like the Bionic Pancreas can adapt to those variations in insulin needs automatically every five minutes – in essence, personalized insulin delivery!
The Bionic Pancreas has some other exciting adaptation advantages. Only body weight information is needed to begin dosing insulin and glucagon – the system quickly adapts within a day to whatever a specific person needs. The team has also worked to eliminate carb counting, as patients must only answer a simple question: “Is this a typical, less than typical, or greater than typical meal?” The Bionic Pancreas learns over time from its performance and can change its dosing if blood sugars are running consistently high (e.g., sickness) or low (e.g., lots of exercise while on vacation).
How does Beta Bionics’ timeline compare to other automated insulin delivery companies?
The current timeline puts Beta Bionics ~18 months behind Medtronic’s MiniMed 670G insulin-only hybrid-closed loop system, which recently finished its pivotal trial and will be submitted to the FDA before the end of this June (potential launch by April 2017). TypeZero’s “inControl” system – the commercial version of the UVA DiAs automated insulin delivery system – is also potentially ahead of Beta Bionics, assuming its major trial starting this year can support an FDA approval. The iLet is on a roughly similar timeline to Bigfoot Biomedical, Tandem, and Insulet, who all have pivotal studies planned for 2017.
How much funding does Beta Bionics need?
Beta Bionics needs an estimated $35 million to get through its pivotal trial. In addition to Lilly’s $5 million investment, Beta Bionics has submitted a proposal to fund a large portion of its pivotal study through an NIH grant for major artificial pancreas studies (up to $16.5 million). To date, the Bionic Pancreas team has done everything since its first grant in 2006 on a tiny budget of $18.5 million – a huge testament to the team’s productivity and incredible drive.
Insulin-only or insulin+glucagon?
Ultimately, we believe that the question is partially one of patient preferences. There will be some patients who may want the extra glycemic control, hypoglycemia protection, and spontaneity offered by the addition of glucagon. The Bionic Pancreas with glucagon could be especially helpful for those with hypoglycemia unawareness, a sizeable percentage of patients. On the other hand, cost considerations could present a barrier to adoption, as the dual-hormone version of the Bionic Pancreas does add the cost of glucagon and a dual-chambered pump, and potentially a custom infusion set (though it is unknown if this infusion set would increase costs). As noted above, the iLet can function as an insulin-only system, meaning its costs without glucagon could be similar to other automated insulin delivery systems.
We believe a range of options is a great thing for people with diabetes, since all systems and products have pros and cons. It’s hard to know at this point how the final commercialized devices will exactly compare, though it will be exciting to follow this field throughout 2016-2018 and beyond. Access remains, of course, one of the very most important questions – and one that the entire Beta Bionics team is very focused on. For a technology to drop A1c by a point or more, as well as reduce hypoglycemia as significantly as it has in trials thus far, this system does something we hope will impress payers as much as it has patients and families (see here for our personal experience with the system).
Photo Credit: betabionics.com